Selling unit: per 100ml
Product Name
Common name: Fibrinogen degradation product (FDP) assay kit (immunoturbidimetric method)
English name: Fibrin/Fibrinogen Degradation Products (FDP) Assay Kit
(Immunoturbidimetric Method)
Reagent Ratio
3:1, 4:1, 5:1, common ratio 3:1, other ratios need to be customized
Intended Use
This kit is used for the in vitro quantitative determination of fibrin(pro)degradation products in serum or plasma.
Fibrin degradation products mainly reflect fibrinolytic function. Increased is seen in: a) primary hyperfibrinolysis; b) secondary hyperfibrinolysis: hypercoagulable state, diffuse intravascular coagulation, renal disease, organ transplant rejection, thrombolytic therapy, etc.; c) vascular embolic diseases (pulmonary embolism, myocardial infarction, occlusive cerebrovascular disease, deep venous thrombosis); d) post-chemotherapy induction phase in leukemia, hemorrhagic thrombocytosis uremia, liver disorders, etc.
Test Principle
The FDP in the sample reacts with the FDP antibody encapsulating the latex particles to form turbidity, resulting in an increase in absorbance, and the change in absorbance is detected at 600 nm and the degree of change is proportional to the concentration of FDP.
Main Components
Reagent components included in the product.
*Components are not interchangeable in kits with different lot numbers.
*Reagent components not included in the product, but necessary for the test: outsourced normal and abnormal QCs and calibrators.
Storage Conditions And Expiration Date
The kit is stored at 2-8℃ away from light and is valid for one year; reagents that have been opened are noted to avoid contamination.
Reagents R1 and R2 are stable for one month when opened and stored at 2-8°C away from light.
Applicable Instruments
Hitachi 7180/7600; Olympus AU680/2700/5421; Toshiba TBA120/40; Myriad BS300/400; Siemens ADVIA 1800/2400 and other automatic and semi-automatic biochemical analyzers.
Sample Request
- Please use plasma (citric acid, potassium EDTA or heparin anticoagulation), or serum collected from blood collection tubes with antifibrinolytic and procoagulant agents. Blood should be centrifuged and separated immediately after collection and tested on the same day if possible. If the sample cannot be tested on the same day, it can be stored at 4-8°C for one day and at -80°C for one month. In addition, the thawed sample can only be used once; repeated freezing and lysing will affect the assay value.
- When injecting specimens and standards, etc., be careful not to create air bubbles.
- Specimens containing insoluble material should be centrifuged or filtered to remove insoluble material before use.
- Specimens with high concentrations beyond the measurement range need to be diluted with a separately available diluent before determination.
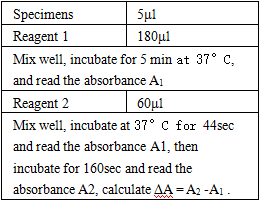
Test Method
- Reagent preparation: liquid reagents are ready to use out of the bottle.
- test conditions: (different parameters on the machine can be requested according to different testing instruments)
Operation steps.
*Reagents and sample volumes can be increased or decreased in proportion to the requirements of different biochemical analyzers.
- Calibration procedure.
The standard curve is plotted with △A as the vertical coordinate and concentration as the horizontal coordinate, and the standard curve is corrected by the non-linear method Spline mode, and this standard curve is saved for calculating the results. The reagents of different batches must be re-calibrated.
- quality control control procedures
Select a QC with the calibrator and the QC measurement value should be within the range of the indicated value. If the results deviate from the range, find the cause by following the steps below.
5.1 Check whether the parameter settings and light source are correct.
5.2 Check that the colorimetric cup and aspiration needle are clean.
5.3 Check the water for contamination, bacterial growth can lead to incorrect results.
5.4 Check the reaction temperature.
5.5 Check the expiration date of the kit.
- Calculation.
Sample concentration = (sample ΔA/min)/(calibration ΔA/min) × calibration concentration
Positive Judgment Value Or Reference Interval
<5μg/ml.
It is recommended that each laboratory establish its own reference range of normal values.
Interpretation Of Test Results
Professional staff is responsible for the review of test results, the analysis of test results, subject to age, gender, diet, geographical influence, usually within the reference range considered normal, such as beyond the range, should be re-measured for confirmation, test results such as the appearance of clinical inconsistency or even contradictory, should be analyzed to find the cause.
Limitations Of The Test Method
Rheumatoid factor ≤200IU/ml, bilirubin ≤20mg/dl, intralipid ≤600mg/dl and hemoglobin concentration ≤500mg/dl had basically no effect on the measured values.
Product Performance Index
Absorbance of reagent blank: wavelength 600nm, optical diameter 1.0cm, temperature 37℃, A≤2.0.
Analytical sensitivity: When the kit tests 20μg/ml of the analyte, the absorbance change rate ΔA/min is between 0.0100 and 0.1000.
Linearity interval: In the interval from 2.5 to 80 μg/ml, the linear correlation coefficient r≥0.99, in the interval from 2.5 to 15 μg/ml, the absolute deviation of linearity should not exceed 1.5 μg/ml, and in the interval from (15 to 80 μg/ml, the relative deviation of linearity should not exceed ±±10%.
Precision: repeatability CV≤10%; relative extreme difference between batches≤10%.
Accuracy: Relative deviation ≤ 10%.
Caution
- The reagent contains sodium azide, please rinse thoroughly with water immediately if you accidentally get it in your eyes or mouth or on your skin, and go to the hospital if necessary.
- The reagent contains sodium azide, which can react strongly with copper, lead and other metals to form azide metal, so please fully dilute the waste solution and flush the drain when discarding to avoid residue in the drain.
- Do not mix or exchange reagents with different lot numbers during the testing process.
- Opened reagents should be stored airtight according to the designated method, and their use is prohibited after the expiration date.
- Please dispose of test tubes and other instruments that have come into contact with test specimens in accordance with the relevant medical waste disposal regulations.
- Although the raw material has been tested for HIV (HIV1, HIV2) antibodies, hepatitis B surface antigen (HbsAg) and hepatitis C virus (HCV) antibodies and found to be negative, although the assay is highly accurate and cannot guarantee that all infected donors will be detected, the QC should also be treated as an infectious specimen.
Reference
Wang M.S., Lu H., Pan J.Y., et al. Changes in antithrombin and fibrinolytic function during liver transplantation. Zhejiang Laboratory Medicine, 2005,3(2):38-40