Selling unit: per 100ml
Homocysteine is an intermediate amino acid formed during the conversion of methionine to cysteine. Homocystinuria is a rare autosomal recessive disorder characterised by a large increase in plasma and urine homocysteine concentrations. Clinical manifestations of homocysteinuria include developmental delay, a malfunctioning equine syndrome-like appearance, osteoporosis, ocular abnormalities, thromboembolic disease and severe premature atherosclerosis of the arteries.
The Homocysteine Reagent is a rapid quantitative assay for homocysteine concentrations in blood, urine and various biological samples. The reagents are fast, accurate, reproducible and traceable and can be used in a wide range of biochemical analysers, POCT instruments and in clinical laboratories, food testing and research.
The reagents have been tested for stability by repeated freeze-thawing and shaking, and have been tested at 37°C and 45°C. The reagents can be transported over long distances at room temperature and can be easily transported around the world.
Product Name
Common name: Homocysteine (HCY) assay kit (circular enzyme method)
English name: HCY Reagent Kit (Enzymatic Cycling Method)
Reagent Ratio
4:1,10:1, other ratios need to be customized
Intended Use
This kit is used to determine the amount of homocysteine (HCY) in human serum or plasma.
Homocysteine (Hcy) is a sulfur-containing amino acid produced by methionine metabolism. 80% of Hcy is bound to proteins in the blood through disulfide bonds, and only a small percentage of free homocysteine is circulating. It is an important risk factor for the development of cardiovascular disease. Increased Hcy in the blood causes damage to the arterial vessels due to irritation of the vessel walls, leading to inflammation and plaque formation in the vessel walls, and ultimately to obstruction of blood flow to the heart. In patients with hyperhomocysteinuria, a severe genetic defect affects Hcy metabolism, resulting in hyper-Hcy blood, and B-vitamin deficiency is associated with moderate or mild Hcy elevation, which also increases the risk of heart disease.
Storage Conditions And Expiration Date
The kit is stored at 2-8°C away from light and is valid for one year.
Reagents that have been opened are careful not to be contaminated, and reagents are stable in the instrument compartment (2-8°C) for one month.
The reagents must not be frozen.
Applicable Instruments
Hitachi 7180/7600; Olympus AU680/2700; Toshiba TBA120; Myriad BS2000M/480; Siemens ADVIA 1800/2400 series automatic biochemical analyzers.
Sample Request
Samples are fresh serum and plasma (heparin anticoagulated). Samples are stable at room temperature for 4 d, at 2-8°C for several weeks, and at -20°C for several months or years.
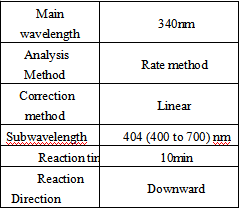
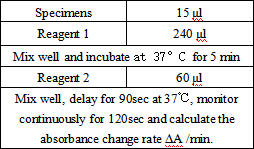
Test Method
- Reagent preparation: liquid reagents are ready to use out of the bottle.
- test conditions: (different test instruments can be requested according to the different parameters on the machine)
Operation steps.
Reagents and sample volumes can be increased or decreased in proportion to the requirements of different biochemical analyzers.
- Calibration procedure.
The use of matching calibrators is recommended.
- QC control procedures.
It is recommended that each laboratory establish its own quality control system and select appropriate quality control products for quality control. The measured values of QC products should be within the specified range. If outside the specified range, it is necessary to take appropriate measures or contact the manufacturer.
- Calculation.
HCY content in the sample (μmol/L) =(ΔAsample – ΔAblank ) × Cstandard / (ΔAstandard – ΔAblank )
Positive Judgment Value Or Reference Interval
Adult: ≤15 μmol/L
It is recommended that each laboratory establish its own reference range of normal values.
Interpretation Of Test Results
The determination of HCY in serum or plasma is only one of the indicators used by the clinician to diagnose the patient. The clinician must also make a comprehensive judgment based on the patient’s physical symptoms, medical history, and other diagnostic items and tools.
Limitations Of The Test Method
Ascorbic acid ≤ 30 mg/dl, bilirubin ≤ 20 mg/dL, hemoglobin ≤ 500 mg/dL, and celiac disease ≤ 500 mg/dL did not interfere with the determination.
Product Performance Index
Absorbance of reagent blank: wavelength 340 nm, optical diameter 1.0 cm, temperature 37°C, A0 ≥ 1.0.
Absorbance change rate of reagent blank: △A/min≤0.015 at wavelength 404 nm and optical diameter 1.0 cm.
Analytical sensitivity: The kit tests 10 μmol/L of the test substance with absorbance change rate ΔA/min ≥ 0.01.
Linearity range: test serum samples, reagent linearity in the interval of 1~50 μmol/L, linear correlation coefficient|r| should not be less than 0.975; in the interval of 1~10 μmol/L, the absolute deviation of linearity should not exceed 1 μmol/L; in the interval of (10~50 μmol/L, the relative deviation of linearity should not exceed ±10%.
Precision: repeatability CV ≤ 5%; relative extreme difference between batches ≤ 8%.
Accuracy: Relative deviation ≤ 10%.
Caution
- This product is for in vitro diagnosis only.
- Avoid contamination when using the reagent, the container used must be clean, and please take necessary precautions, do not swallow, and avoid contact with skin and mucous membrane.
- Please dispose of the measured samples and waste liquids in accordance with the relevant national and local laws and regulations.
- Do not mix R1 and R2 into a single reagent.
- When changing the reagent lot number, please recalibrate.
Reference
- McCully KS. Vascular pathology of homocysteinemia implications for the pathogenesis of arteriosclerosis Am J pathol. (1969) :56:111-128
- McCully KS Chemical pathology of homocysteine: I. Atherogenesis. Ann Clin Lab Sei.(1993);23;447-493
- Cramer DA. Hcmocysteine vs choiesterol-competing views, or a unifying explanation of arteriosclerotic cardiovascular diease Laboratory Medicinevol 29 ,no.7 July (1998).