Selling unit: per 100ml
Product Name
- Common name: Inorganic phosphorus (P) assaykit (UV spectrophotometric method)
- English name: P Reagent Kit (UV Spectrometric Method)
Reagent Ratio
Single reagent
Intended Use
Used to determine the amount of P in human serum, plasma or urine.
Elevated serum P is commonly associated with impaired phosphate excretion in advanced chronic nephritis, hypoparathyroidism, and excessive vitamin D intake; decreased serum P is commonly associated with chondromalacia, rickets, hyperparathyroidism, diabetic coma, and renal tubular absorption dysfunction.
Test Principle
Under acidic conditions, P interacts with molybdate to produce phosphomolybdate with light absorption characteristics. The absorbance increase of the reaction solution is measured at 340 nm and compared with the absorbance increase of the standard solution, the content of P in the sample can be measured.
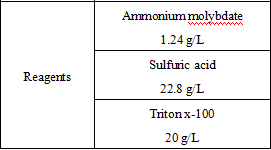
Main Components
1 For the reagent components included in the product.
*Components are not interchangeable in kits with different lot numbers.
*Reagent components not included in the product, but necessary for the test: outsourced normal and abnormal QCs and calibrators.
Storage Conditions And Expiration Date
The kit is stored at 2-8°C away from light and is valid for one year.
Reagents that have been opened are taken care not to be contaminated and the reagents are stabilized in the instrument compartment (2-8°C) for one month.
The reagents must not be frozen.
Applicable Instruments
Hitachi 7180/7600; Olympus AU680/2700; Toshiba TBA120; Myriad BS2000M/480; Siemens ADVIA 1800/2400 series automatic biochemical analyzers.
Sample Request
Hemolysis should be avoided for both serum or plasma specimens, and blood samples taken on an empty stomach are more appropriate. Fluoride anticoagulation can falsely reduce the assay results and heparin resistance or EDTA coagulation of plasma samples is recommended. The urine sample should be dissolved before measurement. 24-hour urine samples should be acidified with 15 ml of concentrated hydrochloric acid. Urine samples should be diluted 1:20 before measurement. Serum or plasma samples can be stored at room temperature (15~25℃) for 24h, 2~8℃ for 4 days, and frozen (-20℃) for 12 months.
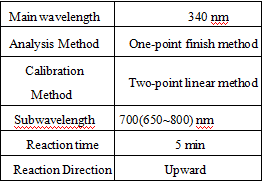
Test Method
- Reagent preparation: liquid reagents are ready to use out of the bottle.
- test conditions that must be met: (different test instruments can be requested according to the different parameters on the machine)
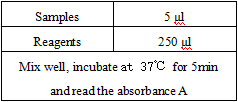
Operating steps.
*Reagents and sample volumes can be increased or decreased in proportion to the requirements of different biochemical analyzers.
- Calibration procedure.
Randox calibration sera are recommended and the method of choice for Randox calibration sera is the UV phosphate method.
- QC control procedures.
It is recommended that each laboratory establish its own quality control system and select appropriate quality control products for quality control. The measured values of QC products should be within the specified range. If outside the specified range, it is necessary to take appropriate measures or contact the manufacturer.
- Calculation of test results.
P concentration in the sample (mmol/L) =(ΔAsample – ΔAblank ) × Cstandard / (ΔAstandard – ΔAblank )
Positive Judgment Value
Adult serum and plasma: 0.81 ~ 1.65 mmol/L
Adult 24h urine: 12 ~ 42 mmol/24h
It is recommended that each laboratory establish its own range of reference values.
Interpretation Of Test Results
The measurement of P in serum, plasma or urine is only one of the indicators used by the clinician to make a diagnosis of the patient. The clinician must also make a comprehensive judgment based on the patient’s physical symptoms, medical history and other diagnostic items and diagnostic tools.
Limitations Of The Test Method.
Bilirubin ≤ 20 mg/dL, hemoglobin ≤ 100 mg/dL and celiac disease ≤ 500 mg/dL did not interfere with the assay.
Product Performance Index
Absorbance of reagent blank: wavelength 340 nm, optical diameter 1.0 cm, temperature 37°C, A0 ≤ 0.5.
Analytical sensitivity: The kit tests 6.5 mmol/L of the analyte, the absorbance change value ΔA ≥ 0.05.
Linearity interval: test serum samples, reagent linearity 0.2 ~ 4.0 mmol/L interval, linear correlation coefficient |r| should not be less than 0.990; in the 0.2 ~ 1.0 mmol/L interval, the absolute deviation of linearity should not exceed 0.1 mmol/L; in the (1.0 ~ 4.0 mmol/L interval, the relative deviation of linearity should not exceed ± 10%.
Precision: repeatability CV ≤ 6%; relative extreme difference between batches ≤ 8%.
Accuracy: Relative deviation ≤ 10%.
Caution
- 1. This product is for in vitro diagnosis only.
- 2. Avoid contamination when using the reagent, the container used must be clean, and please take necessary precautions, do not swallow, and avoid contact with skin and mucous membrane.
- 3. Please dispose of the measured samples and waste liquids in accordance with the relevant national and local laws and regulations.
- When changing the reagent lot number, please recalibrate.
- Samples such as hemolysis, lipidemia and jaundice may cause false elevation of the assay results, which can be corrected by using a sample blank tube: add 5 μl of sample to 250 μl of physiological saline. After mixing, zero the sample at 340 nm with saline, read the absorbance value of the sample blank tube, and subtract the absorbance value of the sample blank tube from the absorbance value of the sample tube.
Reference
- Wang J., Chen C. C., Osaki S., Clin. Chem, (1983); 29: 1255
- Daly J. A. And Ertingshausen G., Clin. Chem, (1983); 18: 263