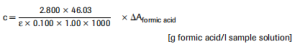Selling unit: per 100ml
Principle
Oxalic acid (oxalate) is cleaved to formic acid (formate) and CO2 at pH 5.0
in the presence of oxalate decarboxylase (1).
The formate formed is quantitatively oxidized to bicarbonate by
nicotin- amide-adenine dinucleotide (NAD) at pH 7.5 in the presence of the enzyme formate dehydrogenase (FDH) (2).
The Test-Combination contains
Bottle 1 with approx. 3 ml solution, consisting of: potassium phosphate/citrate buffer, pH approx. 5.0
Bottle 2 with approx. 0.5 ml oxalate decarboxylase suspension, approx. 8 U
Bottle 3 with approx. 45 ml solution, consisting of: potassium phosphate buffer, 0.1 M; pH approx. 9.5
Bottle 4 with approx. 420 mg lyophilized NAD, Li-salt
Bottle 5 with formate dehydrogenase lyophilizate, 80 U
Bottle 6 with oxalic acid assay control solution for assay control purposes (measurement of the assay control solution is not necessary for calculat- ing the results.) Use the assay control solution undiluted. (Expiry date: see pack label)
Preparation of solutions
Use contents of bottles 1 and 2 undiluted.
Dissolve contents of bottle 4 with the buffer solution from bottle 3 while stirring with a magnetic stirrer (= reagent mixture 4).
Dissolve contents of bottle 5 with 1.2 ml redist. water (= solution 5).
Stability of reagents
Solution 1 is stable at 2–8°C (see pack label).
Bring solution 1 to 20–25°C before use.
The contents of bottle 2 are stable at 2–8°C (see pack label).
Bring suspension 2 to 20–25°C before use.
The contents of bottles 3 and 4 are stable at 2–8°C (see pack label) .
Reaction mixture 4 is stable for 2 weeks at 2–8°C. Bring reaction mixture 4 to 20–25°C before use.
The contents of bottle 5 are stable at 2–8°C (see pack label).
Solution 5 is stable for 5 days at 2–8°C or for 6 weeks at —15°C to —25°C, respectively.
Procedure
Wavelength: 340 nm, Hg 365 nm or Hg 334 nm
Glass cuvette: 1.00 cm light path
Temperature: 20 –25°C
Final volume: 2.800 ml
Read against air: (without a cuvette in the light path), against water oragainst sample blank
Sample solution: 0.8–40 µg oxalic acid/assay4 (in 0.100-0.500 ml sample volume)
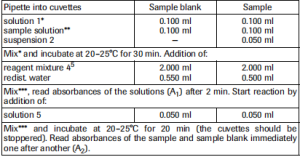
* Pipette solution 1, suspension 2 and sample solution, each onto the bottom of the cuvette, mix by swirling. When using a plastic spatula, remove it from the cuvette only directly before measuring absorbance A1.
** Rinse the enzyme pipette or the pipette tip of the piston pipette with sample solution before dispensing the sample solution.
*** For example, with a plastic spatula or by gentle swirling alter closing the cuvette with Para- film (trademark of the American Can Company, Greenwich, Ct., USA)
Determine the absorbance differences (A2—A1) for both, sample blank and sample. Subtract the absorbance difference of the sample blank from the absorbance difference of the sample.
For each sample a sample blank has to be carried out.
The measured absorbance differences should, as a rule, be at least 0.100 absorbance units to achieve sufficiently precise results (see “Instructions for performance of assay”, and “Sensitivity and detection limit”, pt. 4.)
Calculation
According to the general equation for calculating the concentrations:
If the sample has been diluted during preparation, the result must be multi- plied by the dilution factor F.
When analyzing solid and semi-solid samples which are weighed out for sample preparation, the result is to be calculated from the amount weighed:
- Instructions for performance of assay
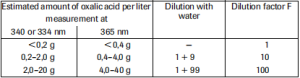
The amount of oxalic acid present in the assay has to be between 1.5 µg and 40 µg (measurement at 365 nm) or 0.8 µg and 20 µg (measurement at 340, 334 nm) respectively. In order to get a sufficient absorbance difference, the sample solution is diluted to yield an oxalic acid concentration between 0.07 and 0.4 g/I or 0.04 and 0.2 g/I, respectively.
Dilution table
If the measured absorbance difference (ΔA) is too low (e.g. < 0.100), the sample solution should be prepared again (weigh out more sample or dilute less strongly) or the sample volume to be pipetted into the cuvette can be increased up to 0.500 ml. The volume of water added must then be reduced so as to obtain the same final volume in the assays for sample and blank (in this case solution 1 is not necessary.) The new sample volume v must be taken into account in the calculation.
Technical information
Oxalic acid and its salts have to be released from solid samples (e.g. fruit, vegetable, baby food) by boiling with hydrochloric acid (1 M) during sample preparation.
Solution 1 and the enzyme suspension 2 are pipetted (e.g. with a piston-type pipette) onto the bottom of the cuvette. Subsequently the sample solution is pipetted onto the surface of the solution in the cuvette (and not onto the wall of the cuvette). (One must not be afraid of a contamination, because a new pipette (tip) is used for the next sample solution.)
Jerky swinging or swirling of the cuvette proved to be best when mixing small volumes (e.g 0.250 ml) in the cuvette.
In carrying out the calculation, a clear indication should be given as to whether the results are to be given as oxalic acid (molar mass
90.04 g/mol) or as oxalate (molar mass 88.02 g/mol). (In enzymatic determinations, the oxalate ion is measured.)
Specificity
The method is specific for oxalic acid.
In the analysis of commercial oxalic acid dihydrate (molecular weight 126.1), results of < 100% have to be expected because the material absorbs mois- ture.
Sensitivity and detection limit
The smallest differentiating absorbance for the procedure is 0.005 absor- bance units. This corresponds to a maximum sample volume v = 0.500 ml and measurement at 340 nm of an oxalic acid concentration of 0.4 mg/I sample solution (if v = 0.100 ml, this corresponds to 2 mg/I sample solution).
The detection limit of 1.6 mg/I is derived from the absorbance difference of
0.020 (as measured at 340 nm) and a maximum sample volume v = 0.500 ml.
Linearity
Linearity of the determination exists from 0.8 µg oxalic acid/assay (1.6 mg oxalic acid/I sample solution; sample volume v = 0.500 ml) to 40 µg oxalic acid/assay (0.4 g oxalic acid/I sample solution; sample volume v = 0.100 ml).
Precision
In a double determination using one sample solution, a difference of 0.005 to
0.010 absorbance units may occur. With a sample volume of v = 0.100 ml and measurement at 340 nm, this corresponds to an oxalic acid concentra- tion of approx. 2–4 mg/I. (If the sample is diluted during sample preparation, the result has to be multiplied by the dilution factor F. If the sample is weighed in for sample preparation, e.g. using 1 g sample/100 ml = 10 g/I, a difference of 0.02–0.04 g/100 g can be expected.)
The following data for the determination of oxalic acid have been published in the literature:
Tomato juice:
x = 21.3 mg/I s = 1.24 mg/I CV = 5.83 %
x = 127.5 mg/I s = 2.89 mg/I CV = 2.27 %
x = 256.0 mg/I s = 1.89 mg/I CV = 0.74 %
Interference
Reducing substances in the sample may interfere with the assay because the reaction rate of formate dehydrogenase is decreased. The reaction time is approx. 40 min when 10 µg sulfur dioxide is present in the assay mixture. The influence of SO2 can be removed completely by addition of 10 µl of hydrogen peroxide (30% w/w) to the assay.
L-Ascorbic acid may interfere with the determination of oxalic acid by formation of complexes. In this case it is recommended to remove the L-ascorbic acid by adding of 17 U ascorbate oxidase6/ml sample solution or by using an ascorbate oxidase spatula7.
The formate dehydrogenase reaction (reaction 2) is delayed in the pres- ence of formaldehyde. Small quantities (5 µg/assay) reduce the speed of the reaction; 10 µg of formaldehyde inhibit the reaction slightly, 100 µg very strongly. Cleavage of oxalic acid by oxalate decarboxylase is not interfered with formaldehyde.
It has not been observed that amines (e.g. ingredients of fish products) interfere with the assay.
Recognizing interference during the assay procedure
If the conversion of formic acid has been completed according to the time given under “Procedure”, it can be concluded in general that no interference has occurred.
On completion of the reaction, the determination can be restarted by adding formic acid or formate (qualitative or quantitative): if the absorb- ance is altered subsequent to the addition of the standard material, this is also an indication that no interference has occurred.
The reaction cannot be restarted with oxalic acid as, subsequent to alter- ing the reaction conditions from pH 5.0 to pH 7.5 (“change of the buffer”), oxalic acid is no longer cleaved.
Operator error or interference of the determination through the pres- ence of substances contained in the sample can be recognized by carrying out a double determination using two different sample volumes (e.g. 0.100 ml and 0.200 ml): the measured differences in absorbance should be proportional to the sample volumes used.
When analyzing solid samples, it is recommended that different quanti- ties (e.g. 1 g and 2 g) be weighed into 100 ml volumetric flasks. The absorbance differences measured and the weights of sample used should be proportional for identical sample volumes.
The use of “single” and “double” sample volumes in double determina- tions is the simplest method of carrying out a control assay in the determi- nation of oxalic acid.
Possible interference caused by substances contained in the sample can be recognized by using an internal standard as a control: in addition to the sample, blank and standard determinations, a further determina- tion should be carried out with sample and assay control solution in the same assay. The recovery can then be calculated from the absorbance differences measured.
Possible losses during the determination can be recognized by carrying out recovery tests: the sample should be prepared and analyzed with and without added standard material. The additive should be recovered quantitatively within the error range of the method.
Reagent hazard
The reagents used in the determination of oxalic acid are not hazardous materials in the sense of the Hazardous Substances Regulations, the Chem- icals Law or EC Regulation 67/548/EEC and subsequent alteration, supple- mentation and adaptation guidelines. However, the general safety measures that apply to all chemical substances should be adhered to.
After use, the reagents can be disposed of with laboratory waste, but local regulations must always be observed. Packaging material can be disposed of in waste destined for recycling.
General Information on sample preparation
In carrying out the assay:
Use clear, colorless and practically neutral liquid samples directly, or after dilution according to the dilution table, and of a volume up to 0.500 ml;
Filter turbid solutions;
Degas samples containing carbon dioxide (e.g. by filtration);
Treat “strongly colored” samples that are used undiluted or with a higher sample volume with activated charcoal or with polyvinylpolypyrrolidone (PVPP), e.g 1–2 g/100 ml;
Crush or homogenize solid or semi-solid samples, extract with water or dissolve in water and filter if necessary;
Deproteinize samples containing protein with trichloroacetic acid;
Extract samples containing fat with hot water (extraction temperature should be above the melting point of the fat involved). Cool to allow the fat to separate, make up to the mark, place the volumetric flask in an ice bath for 15 min and filter.
Application examples
Determination of oxalic acid in fruit and vegetable juices
Adjust pH value of fruit and vegetable juices with potassium hydroxide (2 M) to pH 5.0 before assaying. If L-ascorbate is present in the sample, add approx. 17 U ascorbate oxidase6/ml sample solution, or use 1 ascorbate oxidase spatula7, respectively, mix and incubate for 10 min; use 0.100 ml for the assay.
In the case of colored fruit and vegetable juices, add 100 mg polyvinylpoly- pyrrolidone (PVPP) to 5 ml juice, stir for approx. 1 min and filter; add approx. 100 U ascorbate oxidase afterwards, if necessary.
Adjust strongly acid juices to pH 4.5-5.5 with potassium hydroxide (2 M) if a large sample volume is to be used. Add 17 U ascorbate oxidase/ml, if neces- sary.
Spinach and rhubarb juices should be diluted with redist. water 1 :10 (1 + 9). Use 0.100 ml for the assay.
Determination of oxalic acid in beer
For removing carbonic acid and phenolic substances stir 10 ml of beer with a glass rod in a beaker for 2 min after addition of 0.1 g polyvinylpoly- pyrrolidone (PVPP) and filter through a fluted filter paper. Use the sample, largely free of CO2 and interfering substances, undiluted for the assay (sam- ple volume v = 0.200-0.500 ml).
Determination of oxalic acid in chocolate products
Accurately weigh approx. 5 g of finely grated chocolate or chocolate powder into a 100 ml volumetric flask, add approx. 70 ml of hot redist. water and incubate at 70°C for approx. 15 min. Shake from time to time. Allow to cool to room temperature, fill up to the mark with redist. water and mix.
To separate the fat, place into a refrigerator for approx. 20 min. Filter the cold solution. Discard the first few ml of the filtrate. Use the clear or the slightly opalescent solution for the assay with v = 0.100 ml (maximum 0.200 ml).
For cocoa powder a pretreatment with polyvinylpolypyrrolidone (PVPP) is recommended as stated under determination of oxalic acid in beer.
Determination of oxalic acid in fruits and vegetables
Homogenize fruits and vegetables in a homogenizer. Accurately weigh approx. 100 g of the mixed sample into a beaker, add approx. 300 ml redist. water. Adjust to pH 2–3 (indicator paper) with hydrochloric acid (2 M) and incubate at 50°C for approx. 15 min. Shake from time to time. After cooling to room temperature adjust to pH 5 with potassium hydroxide (2 M), transfer to a 500 ml volumetric flask, fill up to the mark with redist. water and shake vigorously.
Filter the mixture and discard the first few ml. Add 17 U ascorbate oxidase6 to 1.0 ml sample solution to destroy L-ascorbate if present and incubate for 10 min. Take the dilution into account in the calculation. It is also possible to use an ascorbate oxidase spatula7. In this case stir the sample 3 times for 1 min with intervals of 1 min.
Determination of oxalic acid in protein-containing samples
Deproteinize protein-containing samples with trichloroacetic acid (30 mM) in a ratio of 1:3 (1+2), stir for 1 min, neutralize with potassium hydroxide (2 M) and transfer quantitatively into a volumetric flask. Fill up to the mark with redist. water, mix, and filter. Adjust the clear solution, which has to be diluted, if necessary, to pH 5-6 with some drops of hydrochloric acid (2 M), and use it for the assay.
Further applications
The method may also be used in research when analyzing biological samples.
Important note
Keep in mind that the results found will be too low if micro crystals (e.g. calcium oxalate) are present in the sample. It is recommended in these cases to adjust the sample to pH 3.0 with hydrochloric acid (1 M) and to heat the sample in a boiling water-bath for approx. 15 min or to adjust the sample to pH 1.0 and to incubate at room temperature for 1 h.
L-Ascorbate up to a concentration of 100 mg/I sample does not interfere with the assay. Concerning higher concentrations it is recommended to remove the L-ascorbic acid by addition of 17 U ascorbate oxidase6/ml sample solution or by using of one ascorbate oxidase spatula7/ml sample solution.
Determination of formic acid and oxalic acid
If formic acid and oxalic acid are to be determined from one sample solution, proceed as described for procedure. However, a reagent blank has to be performed: pipette 0.100 ml redist. water instead the sample solution.
Determine the absorbance differences (A2—A1) for both, reagent blank and sample. Subtract the absorbance difference of the reagent blank from the absorbance difference of the sample blank.
ΔAformic acid = (A2 — A1)sample blank — (A2 — A1)reagent blank
Calculation:
References
Beutler, H.-O., Becker, J., Michal, G. & Walter, E. (1980) Rapid Method for the Determi- nation of Oxalate, Fresenius Z. Anal. Chem. 301, 186-187
Höpner, T. & Knappe, J. (1974) in Methoden der enzymatischen Analyse (Bergmeyer, H.U.Hrsg.) 3. Aufl., Bd. 2, S. 1596-1600, Verlag Chemie, Weinheim, and (1974) in Methods of Enzymatic Analysis (Bergmeyer, H. U., ed.) 2nd ed., vol. 3, pp. 1551-1555, Verlag Chemie, Weinheim/Academic Press, Inc., New York and London
Brautechnische Analysenmethoden, Band 111, S. 576-580 (1982) Methodensammlung der Mitteleuropäischen Brautechnischen Analysenkommission (MEBAK), herausgege- ben von F. Drawert im Selbstverlag der MEBAK, Freising
Drawert, F., Paul, H. & Hagen, W. (1981) Enzymatische Bestimmung von Oxalsäure und Ameisensäure im Bier, Brauwissenschaft 34, 57-61
Lagemann, M., Graef, V. & Anders, D. (1985) Bestimmung des Oxalsäuregehaltes von Kakao und Kakaoprodukten mit der Oxalat-Decarboxylase Methode, Deutsche Lebens- mittel-Rundschau 81, 140-141
Lagemann, M., Anders, D., Graef, V. & Bödeker, R. H. (1985) Einfluß von Kakao auf die Ausscheidung von Oxalat, Citrat, Magnesium und Calcium im Urin von Kindern, Monatsschr. Kinderheilkd. 133, 754-759