Selling unit: per 100ml
Product Name
Generic name: Low-density lipoprotein cholesterol (LDL-C) assay kit (direct assay)
English name: LDL-C Reagent Kit (Direct Method)
Reagent Ratio
3:1
Intended Use
This kit is used to determine the amount of LDL-C in human serum.
LDL-C has a strong atherosclerosis-promoting effect in serum lipoproteins and is an important risk factor for coronary artery disease. LDL-C is positively correlated with the risk of coronary heart disease. Increased: seen in hyperlipoproteinemia, acute myocardial infarction, coronary artery disease, nephrotic syndrome, chronic renal failure and diabetes mellitus, but also in anorexia nervosa and pregnant women. Decreased: seen in malnutrition, chronic anemia, myeloma, trauma and severe liver disease.
Test Principle
In a cholesterol measurement system in the presence of cholesterol esterase (CHE) and cholesterol oxidase (COD), LDL-C is selectively solubilized by the addition of a specific surfactant to measure LDL-cholesterol. Other lipoproteins (HDL, VLDL, celiac particles) do not react because they are hindered by surfactants and sugar compounds and remain in the reaction solution as lipoproteins. Based on this principle, LDL-cholesterol can be measured directly. The absorbance of the quinone dye at 548 (530-560) nm correlates with the amount of LDL-cholesterol.
Main Components
Reagent components included in the product.

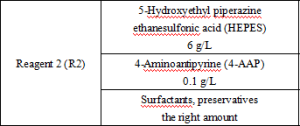
*Components are not interchangeable in kits with different lot numbers.
*Reagent components not included in the product, but necessary for the test: outsourced normal and abnormal QCs and calibrators.
Storage Conditions And Expiration Date
The kit is stored at 2-8°C away from light and is valid for one year.
Reagents that have been opened are careful not to be contaminated, and reagents are stable in the instrument compartment (2-8°C) for one month.
The reagents must not be frozen.
Applicable Instruments
Hitachi 7180/7600; Olympus AU680/2700; Toshiba TBA120; Myriad BS2000M/480; Siemens ADVIA 1800/2400 automatic biochemistry analyzer.
Sample Request
Serum samples.
Patients fast before blood collection, except for bedridden patients, sit still for at least 5 min before blood collection, and take venous blood, the use of tourniquet does not exceed 1 min to avoid hemolysis.
After blood collection, the blood should be left at room temperature for 30-40 min to clot on its own, and the serum should be separated by centrifugation within 1h-2h. The samples should be stored at room temperature for no more than 4h, below 4℃ for 4h-48h, and below -70℃ if longer storage is needed. Do not freeze and thaw repeatedly.
Test Method
- Reagent preparation: liquid reagents are ready to use out of the bottle.
- Test conditions: (the following parameters are set in accordance with Toshiba TBA120, and different on-board parameters can be requested according to different testing instruments)
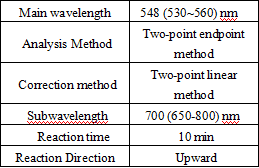
Operation steps.
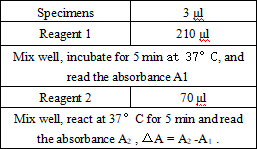
Reagents and sample volumes can be increased or decreased in proportion to the requirements of different biochemical analyzers.
- Calibration procedure.
The use of matching calibrators is recommended.
- QC control procedures.
It is recommended to use quality control sera produced by Randox Laboratories Ltd UK, Landau Laboratory Diagnostics Ltd. It is recommended that each laboratory establish its own QC system and select appropriate QCs for quality control. The QC values should be within the specified range. If outside the specified range, it is necessary to take appropriate measures or contact the manufacturer.
- Calculation.
LDL-C concentration in the sample (mmol/L) =(ΔAsample – ΔAblank ) × Cstandard / (ΔAstandard – ΔAblank )
Reference Range
Ideal level: <3.33 mmol/l ( <129 mg/d L )
Higher than 4.13 mmol/l (160 mg/d L ) is hyperLDL-C .
The reference interval is derived from the third edition of the “National Clinical Inspection Operating Regulations”, and each laboratory should establish its own normal reference range according to the situation of the region and the population.
Explanation Of Test Results
LDL-C in serum is only one of the indicators for clinicians to diagnose patients. Clinicians should also make comprehensive judgments based on the patient’s physical symptoms, medical history, and other diagnostic items and diagnostic methods.
Limitations Of The Test Method
Ascorbic acid ≤ 30 mg/dL , bilirubin ≤ 20 mg/dL , hemoglobin ≤ 500 mg/dL , and chyle ≤ 500 mg/dL did not interfere with the assay.
Product Performance Index
Appearance: The appearance of the kit should be clean, the text symbols should be clear, and the reagent bottle should be intact; R1 is a colorless or light yellow clear liquid, and R2 is a colorless or light yellow clear liquid, without precipitation, suspended matter and floc.
Reagent blank absorbance: wavelength 548 nm , optical diameter 1.0 cm , temperature 37 ℃ , A 0 ≤ 0.05 .
Analytical sensitivity: when the kit tests 1.0 mmol / L analyte, the absorbance change value ΔA ≥ 0.0 3 .
Linearity range: When testing serum samples, the linearity of the reagent is in the range of 0.1 to 11.6 mmol /L , and the linear correlation coefficient |r | mmol/L ; in the range of (3.0 ~ 11.6 mmol/L , the linear relative deviation should not exceed ±10% .
Precision: Repeatability CV≤ 3 % ; Inter-assay relative range ≤ 6% .
Accuracy: Relative deviation ≤ 10% .
Precautions
- This product is only used for in vitro diagnosis.
- Avoid contamination when using reagents, use containers must be clean, and take necessary precautions, do not swallow, and avoid contact with skin and mucous membranes.
- Do not mix R1 and R2 as a single reagent.
- When changing the reagent lot number, please re-calibrate.
- Testing of the same sample using reagents from different manufacturers may vary.
- All sample box reaction waste should be treated as a source of infection, and operators need to take necessary protective measures.
- The reagent contains the preservative sodium azide (toxic). If it gets into the eyes, mouth or gets on the skin, please rinse it thoroughly with water immediately, and go to the hospital if necessary. Sodium azide can react strongly with copper, lead and other metals to form metal azide, so please fully dilute the waste liquid and flush the drain pipe when discarding to avoid residues in the drain pipe.
- Test tubes and other instruments that have been in contact with the test samples should be disposed of in accordance with the medical waste disposal method.
references
- Rifai N, Bachorik PS, Albers JJ. Lipids, lipoproteins and apolipoproteins. In: Burtis CA, Ashwood ER, editors. Tietz Textbook of Clinical Chemistry. 3 rd ed. Philadelphia: WB Saunders Company; (1999). p. 809 -61.
- Recommendation of the Second Joint Task Force of European and other Societies on Coronary Prevention of coronary heart disease in clinical practice. Eur Heart J (1998); 19: 1434-503.




























