Selling unit: per 100ml
Product Name
Common name: Neutrophil gelatinase-associated lipid transport protein (NGAL) assay kit (latex-enhanced immunoturbidimetric assay)
English name: Neutrophil gelatinase-associated lipocalin (NGAL) Assay Kit (Immunoturbidimetric method)
Reagent Ratio
3:1, 4:1, 5:1, common ratio 4:1, other ratios need to be customized
Intended Use
This kit is used for the determination of NGAL in human plasma or urine.
NGAL is not only present in neutrophils, but also in specific epithelial cells, for example, during ischemic and toxic kidney injury, NGAL in tubular epithelial cells will increase significantly, and within the first two hours, NGAL levels in urine and blood will increase significantly, so NGAL can assist in the diagnosis of early acute kidney injury.
High NGAL levels are usually seen in the following situations: patients undergoing cardiovascular surgery, critically ill persons, septic or hemorrhagic shock, renal transplants, reactions to intravenous x-ray contrast agents, and as reflected by nephrotoxic treatment. more than 50% of patients develop some degree of acute renal failure, which usually occurs late in major illnesses making mortality significantly higher.
Test Principle
The NGAL in the sample combines with the anti-human NGAL antibody in the reagent to form an insoluble immune complex, resulting in turbidity of the reaction solution, which is detected at 572 nm and the change in turbidity is proportional to the concentration of NGAL in the sample.
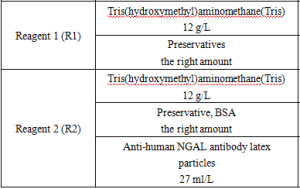
Main Components
Reagent components included in the product.
*Components are not interchangeable in kits with different lot numbers.
*Reagent components not included in the product, but necessary for the test: outsourced normal and abnormal QCs and calibrators.
Storage Conditions And Expiration Date
The kit is stored at 2-8°C away from light and is valid for one year.
Reagents that have been opened are taken care not to be contaminated and the reagents are stabilized in the instrument compartment (2-8°C) for one month.
The reagents must not be frozen.
Applicable Instruments
Hitachi 7180/7600; Olympus AU680/2700; Toshiba TBA120; Myriad BS2000M/480; Siemens ADVIA 1800/2400 series automatic biochemical analyzers.
Sample Request
1, sample type: EDTA or heparin anticoagulated plasma, hemolysis, hyperlipidemia, heat-treated or contaminated specimens will have an effect on the results. The urine specimen was centrifuged within 15 min after collection (2000r/min, 5min) and the supernatant urine was taken for testing.
3、Plasma stored in 2-8℃ can be stable for 1 week, -20℃ can be stable for about 1 month. Urine specimens stored at 4℃ need to be tested within 24 hours, and urine specimens stored at -20℃ can be stored for 7 days.
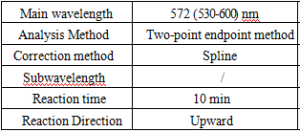
Test Method
- Reagent preparation: liquid reagents are ready to use out of the bottle.
- test conditions: (different test instruments can be requested according to the different parameters on the machine)
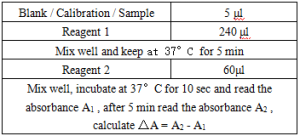
Operation steps.
*Reagents and sample volumes can be increased or decreased in proportion to the requirements of different biochemical analyzers.
- Calibration procedure.
Calibration with matching calibrators is recommended.
- QC control procedures.
It is recommended that each laboratory establish its own quality control system and select appropriate quality control products for quality control. The measured values of QC products should be within the specified range. If outside the specified range, it is necessary to take appropriate measures or contact the manufacturer.
- Calculation.
6-point calibration, input the corresponding value. With △A as the vertical coordinate and concentration as the horizontal coordinate, plot the standard curve, and correct this standard curve by non-linear method Spline, Logit-Log5P and other modes, and save this standard curve for calculating the results. The reagents of different batches must be re-calibrated.
Positive Judgment Value Or Reference Interval
Plasma: 37-180ng/mL
Urine: 0.9-100ng/mL
It is recommended that each laboratory establish its own reference range of normal values.
Interpretation Of Test Results
The determination of NGAL in plasma or urine is only one of the indicators used by the clinician to make a diagnosis of the patient. The clinician must also make a comprehensive judgment based on the patient’s physical symptoms, medical history, and other diagnostic items and diagnostic tools.
Limitations Of The Test Method
Bilirubin ≤ 30 mg/dL and hemoglobin ≤ 500 mg/dL did not interfere with the determination.
Product Performance Index
Absorbance of reagent blank: wavelength 572 nm, optical diameter 1.0 cm, temperature 37°C, A0 ≤ 1.0.
Analytical sensitivity: the kit tests 200 ng/mL of the analyte, the absorbance change value △A ≥ 0.03.
Linearity interval: test serum samples, reagent linearity in the interval of 50-5000 ng/mL, linear correlation coefficient|r| should not be less than 0.990; in the interval of 50-100 ng/mL, the absolute deviation of linearity should not exceed 10 ng/mL; in the interval of (100- 5000 ng/mL, the relative deviation of linearity should not exceed ±10%.
Precision: repeatability CV ≤ 10%; relative extreme difference between batches ≤ 10%.
Accuracy: Relative deviation ≤ 15%.
Caution
- This product is for in vitro diagnosis only and should be rinsed with plenty of water if it comes in contact with human body.
- Newly opened reagents should not be mixed with used reagents, otherwise it is easy to cause the stability of reagents to decrease.
- If the reagent is to be discarded, please dilute it with plenty of water before disposal.
- If the result exceeds the linear range, dilute the specimen 1:1 with physiological saline and multiply the result by 2.
Reference
- Prasad Devarajan. Biomark Med. 2010 April ; 4(2): 265-280. doi:10.2217/bmm.10.12.
- ARASH AGHEL, KEVIN SHRESTHA, WILFRIED MULLENS, ALLENBOROWSKI, and W. H. WILSON TANG.J Card Fail. 2010 January ; 16(1): 49-54. doi: 10.1016/j.cardfail.2009.07.003.