Selling unit: per 100ml
Product Name
- Common name: Apolipoprotein B (ApoB) test kit (immunoturbidimetric assay)
- English name: ApoB Reagent Kit (Immunoturbidimetric Method)
Reagent Ratio
3:1, 4:1, 5:1, common ratio 3:1, other ratios need to be customized
Intended Use
This reagent is used to determine the amount of ApoB in human serum or plasma.
Fats are synthesized in the liver or small intestine, where they are transformed into hydrophilic structures by forming a series of particles before
transported to tissues and organs, these particles are composed of externally wrapped monolayers of proteins (apolipoproteins) and polar lipids (such as
(phospholipids and unlipidated cholesterol) plus an internal neutral lipid core (e.g. triglycerides and cholesterol lipids).
Apolipoproteins play an important role in the further metabolism and catabolism of the above mentioned particles. Apolipoprotein B is a low
The main component of densely packed lipoproteins (LDL), two forms of Apo B have been identified in humans, the most common being
Apo B-100 (or Big B), the lipoprotein in which Apo B is primarily synthesized by the liver. Another form of Apo
Protein B-48 (also known as small B), which is synthesized in the wall of the small intestine, is the main transport protein for cholesterol in the blood, while
It is also the main ligand for intracellular cholesterol uptake into the LDL-receptor, mainly through the LDL-receptor bypass, and Apo B is significantly
It has been shown to have an atherogenic effect and is therefore a major indicator of the risk of coronary heart disease. The results of the current study indicate that Apo
There is a strong relationship between A1 and Apo B and coronary artery disease. Patients with coronary artery disease generally have relatively low Apo A1 levels and relatively high Apo B levels.
Test Principle
ApoB in the serum binds to the specific sheep anti-human ApoB antibody in the reagent to form an insoluble immune complex, causing turbidity in the reaction solution, and the turbidity level reflects the ApoB content in the serum sample.
Main Components
Reagent components included in the product.
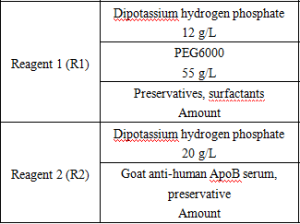
*Components are not interchangeable in kits with different lot numbers.
*Reagent components not included in the product, but necessary for the test: outsourced normal and abnormal QCs and calibrators.
Storage Conditions And Expiration Date
The kit is stored at 2-8°C away from light and is valid for one year.
Reagents that have been opened are careful not to be contaminated, and reagents are stable in the instrument compartment (2-8°C) for one month.
The reagents must not be frozen.
Applicable Instruments
Hitachi 7180/7600; Olympus AU680/2700; Toshiba TBA120; Myriad BS2000M/480; Siemens ADVIA 1800/2400 series automatic biochemical analyzers.
Sample Request
Serum or plasma samples, plasma is anticoagulated with heparin or EDTA. ApoB is stable for 7 days at 2-8°C and for 2 months at -20°C.
Test Method
- Reagent preparation: liquid reagents are ready to use out of the bottle.
- test conditions: (different parameters on the machine can be requested according to different testing instruments)

Operation steps.
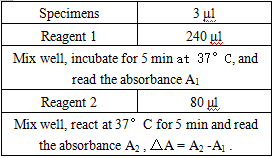
Reagents and sample volumes can be increased or decreased in proportion to the requirements of different biochemical analyzers.
- Calibration procedure.
Landau calibrators are recommended.
- QC control procedures.
It is recommended that each laboratory establish its own quality control system and select appropriate quality control products for quality control. The measured values of QC products should be within the specified range. If outside the specified range, it is necessary to take appropriate measures or contact the manufacturer.
- Calculation.
5 points are calibrated and the corresponding values are entered. With △A as the vertical coordinate and concentration as the horizontal coordinate, the standard curve is plotted, and this standard curve is corrected by the non-linear method Spline, Logit-Log4P and other modes, and this standard curve is saved and used to calculate the results.
Positive Judgment Value Or Reference Interval
0.63~1.14 g/L
It is recommended that each laboratory establish its own reference range of normal values.
Interpretation Of Test Results
Bilirubin ≤ 40 mg/dL, hemoglobin ≤ 500 mg/dL and celiac ≤ 500 mg/dL did not interfere with the determination.
Limitations Of The Test Method
The measurement of ApoB in human serum or plasma is only one of the indicators used by the clinician to make a diagnosis of the patient. The clinician must also make a comprehensive judgment based on the patient’s physical symptoms, medical history, and other diagnostic items and diagnostic tools.
Product Performance Index
Absorbance of reagent blank: wavelength 340 nm, optical diameter 1.0 cm, temperature 37°C, A0 ≤ 0.15.
Analytical sensitivity: the kit tests 1.0 g/L of the analyte, the absorbance change value ΔA ≥ 0.15.
Linearity range: test serum samples, reagent linearity in the interval of 0.30~2.50 g/L, linear correlation coefficient|r| should not be less than 0.975; in the interval of 0.30~0.60 g/L, the absolute deviation of linearity should not exceed 0.06 g/L; in the interval of (0.60~2.50 g/L, the relative deviation of linearity should not exceed ±10%.
Precision: repeatability CV≤ 10.0%; relative extreme difference between batches≤ 10.0%.
Accuracy: Relative deviation ≤ 15%.
Caution
- This product is for in vitro diagnosis only.
- Avoid contamination when using the reagent, the container used must be clean, and please take necessary precautions, do not swallow, and avoid contact with skin and mucous membrane.
- Please dispose of the measured samples and waste liquids in accordance with the relevant national and local laws and regulations.
- For samples with lipid blood visible to the naked eye and triglyceride content exceeding 250 mg/dL, the results need to be diluted with saline before determination and multiplied by the dilution factor.
- Please dispose of the measured samples and waste liquids in accordance with the relevant national and local laws and regulations.
- When changing the reagent lot number, the calibration should be re-calibrated.
Reference
- Bacharik, P. S. et al Clinica Chimica Acta (1988) 30:599, 806
- Albers, J. J. et al Clin Chem (1992); 38: 658
- Albers, J. J. ,Marcovina SM. Kennedy H. International Federation of Clinical Chemistry Standardization Project for measurements of apolipoproteins A-Ⅰand B,ⅡEvaluation and selection of Candidate reference Materials Clin Chem (1992), 38: 658-662.




























