Selling unit: per 100ml
Product Name
Common name: Free fatty acid (NEFA) assay kit (oxidase method)
English name: Non-esterified Fatty Acid (NEFA) Assay Kit (Oxidizing enzyme Method)
Reagent Ratio
3:1,4:1 can be used, other ratios need to be customized, minimum package (R2:1L)
Intended Use
This kit is used for the in vitro quantitative assay of NEFA in human serum or plasma.
Increased NEFA is seen in obesity, diabetes mellitus, myocardial infarction, hyperthyroidism, acromegaly, severe liver disease and starvation; decreased NEFA is seen in hypothyroidism, pituitary insufficiency and Addison’s disease. Physiological decrease can be caused after meals.
Test Principle
This reagent is used to detect NEFA in samples by oxidative enzyme method.
NEFA, in the presence of ATP and coenzyme A, is acted upon by acetyl coenzyme A synthase to produce esteryl coenzyme A. Esteryl coenzyme A is oxidized by acetyl coenzyme A oxidase to produce 2,3-trans-enoyl coenzyme A and hydrogen peroxide, while the generated hydrogen peroxide, in the presence of peroxidase, generates red color with 2-hydroxy-3-m-toluidine propionic acid sodium salt (TOOS) and 4-aminoantipyrine The absorbance of the quinonimine compound varied in proportion to the amount of NEFA in the sample.
NEFA + ATP + CoA ACS Acyl CoA + AMP + PPi
Acyl CoA + O2 ACOD 2,3-trans-Enoyl-CoA + H2 O2
2H2 O2 + chromogen + 4-AAP POD dye + 4H2 O
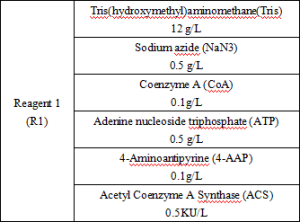
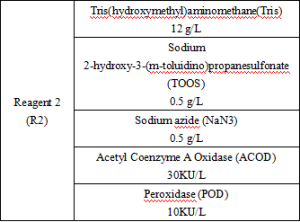
Main Components
Reagent components included in the product.
*The components of the kits are not interchangeable between batches.
*Reagent components not included in the product, but necessary for the test: outsourced normal and abnormal QCs and calibrators.
Storage Conditions And Expiration Date
The kit is stored at 2-8°C away from light and is valid for one year.
Reagents that have been opened are taken care not to be contaminated and the reagents are stabilized in the instrument compartment (2-8°C) for one month.
The reagents must not be frozen.
Applicable Instruments
Hitachi 7180/7600; Olympus AU680/2700; Toshiba TBA120; Myriad BS2000M/480; Siemens ADVIA 1800/2400 series automatic biochemical analyzers.
Sample Request
Serum, plasma.
Use anticoagulants other than heparin (heparin can interfere with the experiment), such as EDTA, sodium citrate, sodium fluoride, ammonium oxalate. Fasting serum samples, which should be analyzed immediately after sample collection and left at room temperature for too long, will have elevated levels of NEFA due to enzyme metabolism.
Test Method
- Reagent preparation: liquid reagents are ready to use out of the bottle.
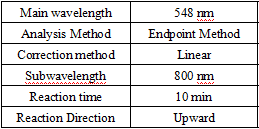
- test conditions: (different test instruments can be requested according to the different parameters on the machine)
Operation steps.
*Reagents and sample volumes can be increased or decreased in proportion to the requirements of different biochemical analyzers.
- Calibration procedure.
Calibration with matching calibrators is recommended.
- QC control procedures.
It is recommended that each laboratory establish its own quality control system and select appropriate quality control products for quality control. The measured values of QC products should be within the specified range. If outside the specified range, it is necessary to take appropriate measures or contact the manufacturer.
Positive Judgment Value Or Reference Interval
Fasting: 0.1-1.05 mmol/l.
Each laboratory should establish its own normal reference range according to the region and population.
Interpretation Of Test Results
The assay of NEFA in serum is only one of the indicators used by the clinician to diagnose the patient. The clinician must also make a comprehensive judgment based on the patient’s physical symptoms, medical history, and other diagnostic items and diagnostic tools.
Limitations Of The Test Method
Ascorbic acid ≤ 25 mg/dL, bilirubin ≤ 25 mg/dL, hemoglobin ≤ 250 mg/dL, and celiac disease ≤ 500 mg/dL did not interfere with the assay.
Product Performance Index
Absorbance of reagent blank: wavelength 548 nm, optical diameter 1.0 cm, temperature 37°C, A0 ≤ 0.2.
Analytical sensitivity: the kit tests 1.00 mmol/L of the test substance, the absorbance change value ΔA ≥ 0.03.
Linearity interval: test serum samples, reagent linearity in the interval of 0.05-3.0 mmol/L, linear correlation coefficient|r| should not be less than 0.990; in the interval of 0.05-0.5 mmol/L, the absolute deviation of linearity should not exceed 0.05 mmol/L; in the interval of (0.5-3.0 mmol/L, the relative deviation of linearity should not exceed ±10% .
Precision: repeatability CV ≤ 5 %; relative extreme difference between batches ≤ 8%.
Accuracy: Relative deviation ≤ 10%.
Caution
- This product is for in vitro diagnosis only.
- The operation process should be well protected.
- As heparin activates lipoprotein esterase, it causes the release of NEFA from the triglycerides carried by apolipoproteins in blood, which skews the results. Therefore, heparin-anticoagulated blood samples, blood samples from patients treated with de-heparin, and heparinized containers are not suitable for this experiment.
- If the result in the sample exceeds 3.0 mmol/L, the sample should be diluted with physiological saline and the result multiplied by the dilution factor. The maximum dilution of this product is 3 times.
Reference.
1 Li Yilong, Zhang Yan. Clinical significance and research progress of free fatty acids in diabetes mellitusJ. International Journal of Laboratory Medicine, 2008, 29(10): 954-956.