Selling unit: per 100ml
Product Name
- Common name: Urea nitrogen (BUN) assay kit (enzyme method)
- English name: BUN Reagent Kit (Enzymic Method)
Reagent Ratio
3:1, 4:1, 5:1 are available.
Intended Use
This kit is used to determine the amount of BUN in human serum, plasma or urine.
BUN is the main end product of protein metabolism in human body, which constitutes most of the non-protein nitrogen in blood, and is produced in liver and excreted to urine through kidney, therefore, the level of BUN depends on the amount of protein intake, protein catabolism and kidney function. The measurement of BUN concentration is important for the course of chronic kidney disease, disease observation and prognosis.
Test Principle
A urease coupled glutamate dehydrogenase reaction was used. Urease first hydrolyzes BUN to produce ammonia and carbon dioxide, and subsequently, in the presence of glutamate dehydrogenase, ammonia combines with α-ketoglutarate to produce glutamate and oxidizes NADH to NAD+ , which causes a decrease in absorbance at 340 nm, which is proportional to the BUN concentration in the sample. By monitoring the rate of decrease in absorbance at 340 nm, the concentration of BUN in the sample can be calculated.
Main Components
Reagent components included in the product.
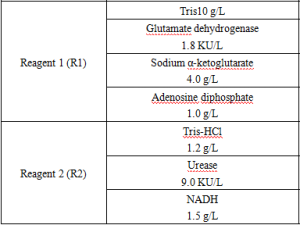
*Components are not interchangeable in kits with different lot numbers.
*Reagent components not included in the product, but necessary for the test: outsourced normal and abnormal QCs and calibrators.
Storage Conditions And Expiration Date
The kit is stored at 2-8°C away from light and is valid for one year.
Reagents that have been opened are taken care not to be contaminated and the reagents are stabilized in the instrument compartment (2-8°C) for one month.
The reagents must not be frozen.
Applicable Instruments
Hitachi 7180/7600; Olympus AU680/2700; Toshiba TBA120; Myriad BS2000M/480; Siemens ADVIA 1800/2400 series automatic biochemical analyzers.
Sample Request
Serum, heparin (ammonium heparin may not be used) or EDTA to anticoagulate plasma and urine. Urea in serum (plasma) samples stored at 2-8°C can be stable for 7 days and -20°C can be stable for 1 year.
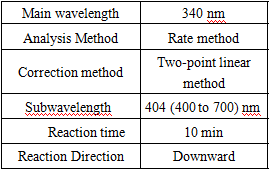
Test Method
- Reagent preparation: liquid reagents are ready to use out of the bottle.
- test conditions: (different test instruments can be requested according to the different parameters on the machine)
Operation steps.
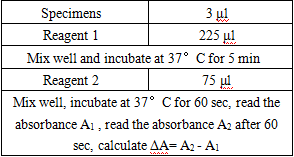
*Reagents and sample volumes can be increased or decreased in proportion to the requirements of different biochemical analyzers.
- Calibration procedure.
Randox calibration sera are recommended. the method of choice for Randox calibration sera is the urease kinetic method.
- QC control procedures.
It is recommended that each laboratory establish its own quality control system and select appropriate quality control products for quality control. The measured values of QC products should be within the specified range. If outside the specified range, it is necessary to take appropriate measures or contact the manufacturer.
- Calculation.
BUN content in the sample (mmol/L) =(ΔAsample – ΔAblank ) × Cstandard / (ΔAstandard – ΔAblank )
Positive Judgment Value
Serum, plasma: 1.7-8.3 mmol/L
Interpretation Of Test Results
The determination of BUN in serum, plasma or urine is only one of the indicators used by clinicians to diagnose patients. Clinicians also have to make a comprehensive judgment based on the patient’s physical symptoms, medical history and other diagnostic items and means.
Limitations Of The Test Method
Bilirubin ≤ 40 mg/dL, hemoglobin ≤ 500 mg/dL and celiac ≤ 500 mg/dL did not interfere with the determination.
Product Performance Index
Absorbance of reagent blank: wavelength:340 nm, optical diameter 1.0 cm, temperature 37°C, A0 ≥ 1.0.
Absorbance change rate of reagent blank: △A/min ≤ 0.01 at wavelength 340 nm and optical diameter 1.0 cm.
Analytical sensitivity: the kit tests 2 mmol/L of the test substance, the absorbance change rate ΔA/min ≥ 0.01.
Linearity interval: test serum samples, reagent linearity 0.2 ~ 35.0 mmol/L interval, linear correlation coefficient |r| should not be less than 0.990; in the 0.2 ~ 9.0 mmol/L interval, the absolute deviation of linearity should not exceed 0.9 mmol/L; in the (9.0 ~ 35.0 mmol/L interval, the relative deviation of linearity should not exceed ± 10%.
Accuracy: Repeatability CV ≤ 6%; relative extreme difference between batches ≤ 10%;
Accuracy: Relative deviation ≤ 10%.
Caution
- 1. This product is for in vitro diagnosis only.
- 2. The reagents should avoid contamination, the containers used must be clean, and the unused reagents should be stored at 2~8℃.
- 3. When changing the reagent lot number, the calibration should be re-calibrated.
- 5. If the reagent is accidentally splashed on human surface such as skin, eyes, etc., it must be flushed with water, and if it is accidentally ingested, it needs to be treated in hospital.
Reference
- Thomas L. Clinical Laboratory Diagnostics. 1sted. Frankfurt:TH-Books Verlagsgesellschaft; (1998). p. 374-7.




























